Features
- Easy to fit, single use lens
- The FIRST and ONLY soft contact lens designed for myopia control and is FDA-approved* to slow the progression of myopia in age-appropriate children†1
- Corrects refractive error and slows the elongation of the eye through ActivControl® Technology
- Suitable for children as young as 8**
- For every MiSight® 1 day‡ lens you fit, we partner with Plastic Bank® to collect and convert an equal amount$ of plastic, which means all MiSight® 1 day contact lenses are now net plastic neutral2.
Learn more about our commitment to sustainability.
The MiSight® 1 day lens is clinically proven to slow the progression of myopia when initially prescribed for children 8-12 years old*. It serves as the cornerstone of a comprehensive myopia management approach.
MiSight® 1 day contact lenses were clinically validated in a multi-year comprehensive study that enrolled children between 8 and 12.
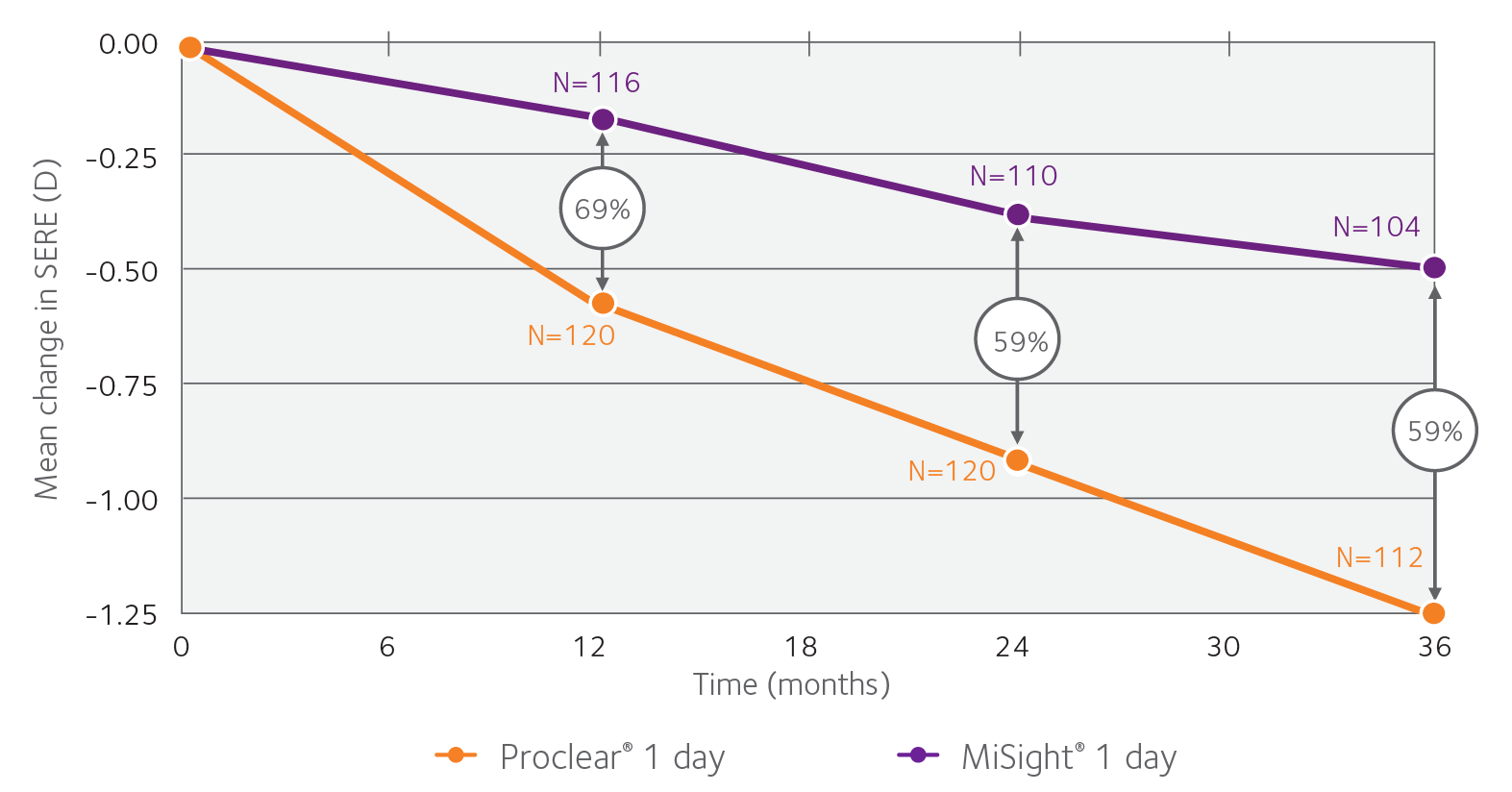
Over three years, MiSight® reduced myopia progression by 59%, versus a single vision 1 day lens.1
The clinical study of MiSight® 1 day lenses was the first to demonstrate sustained reduction in myopia progression with a soft contact lense over a three-year period.1†
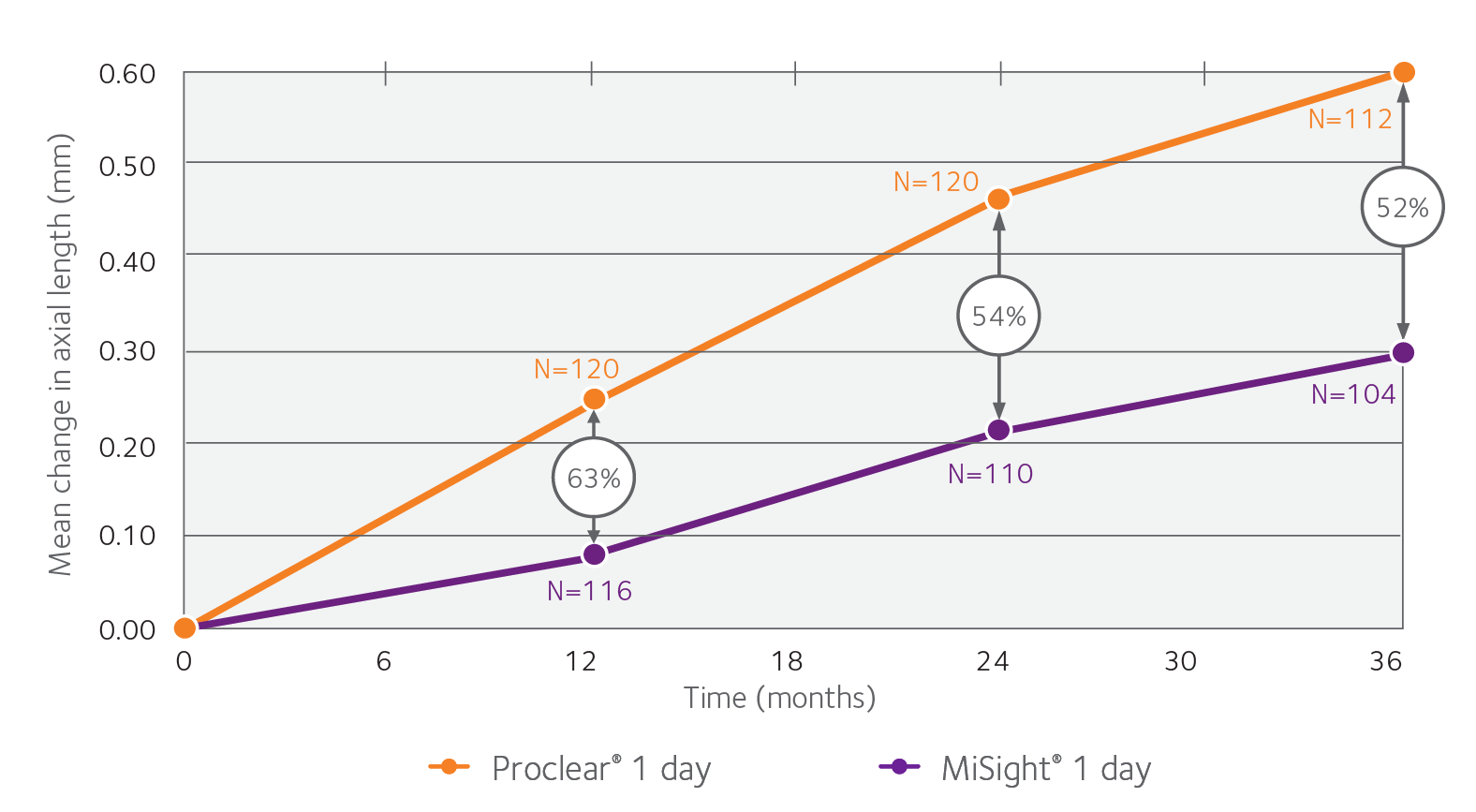
With MiSight®, we also saw 52% reduction in average axial lengthening1†
Change in axial length
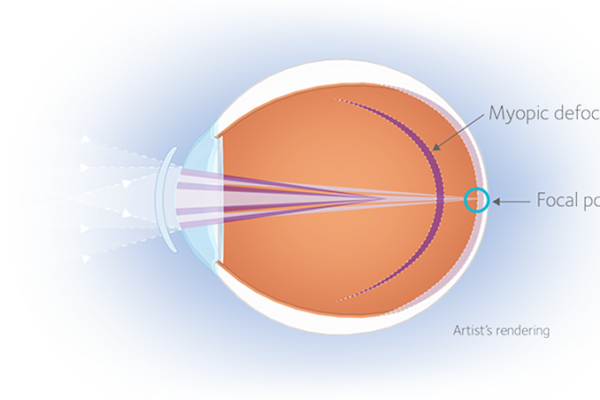
How MiSight® 1 day works
MiSight® 1 day with ActivControl® Technology helps slow the elongation of the eye and myopia progression, while fully correcting refractive error.1 Addressing axial elongation helps to reduce the risk of myopia-related vision complications later in life, including irreversible vision loss.2
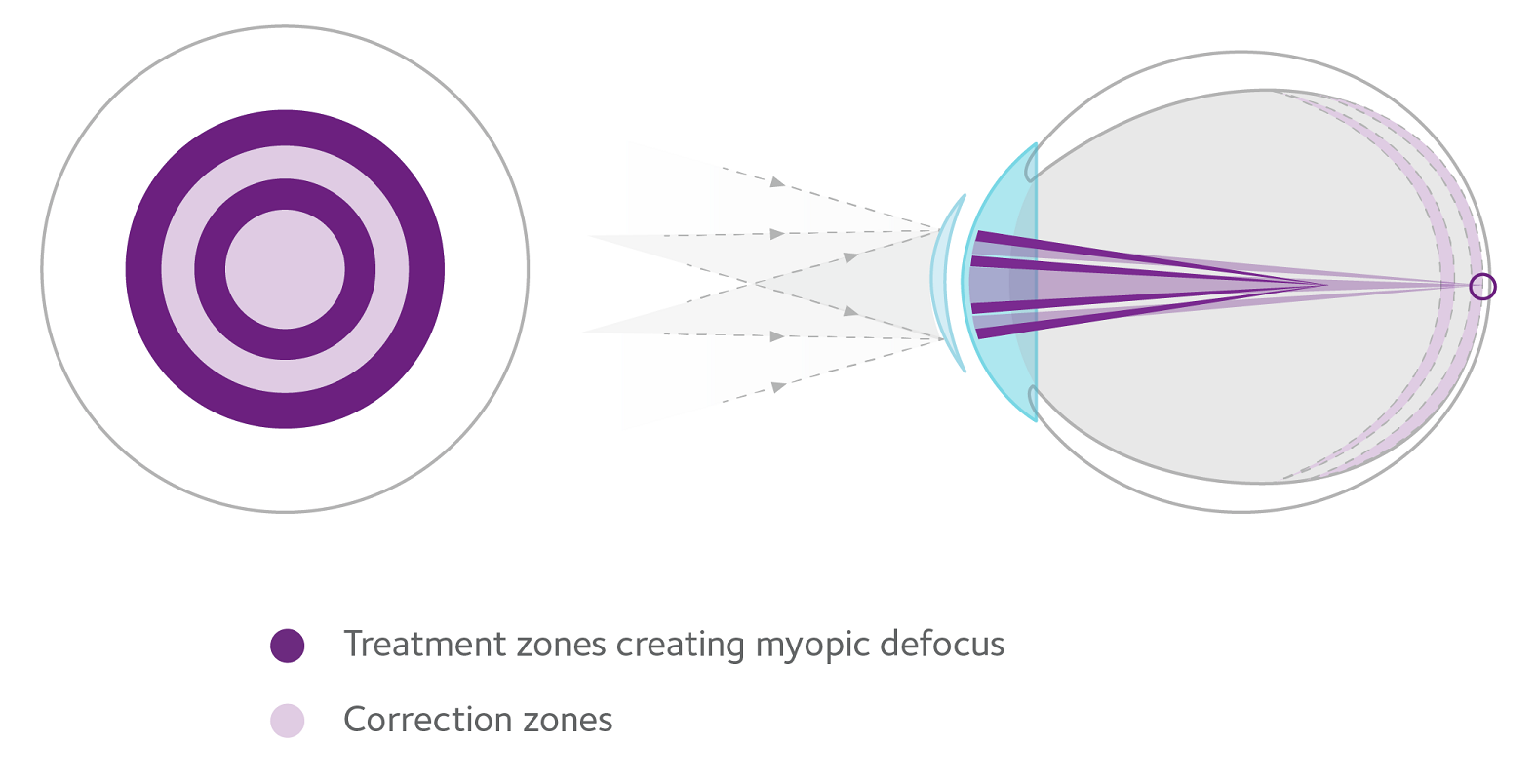
- Two treatment zones create myopic defocus with image focus in front of the retina, rather than behind it to slow axial elongation
- Two correction zones correct myopia in all gaze positions
Lens Technology
Product Details
No Plano
*Indications for Use: MiSight® (omafilcon A) daily wear single use Soft Contact Lenses are indicated for the correction of myopic ametropia and for slowing the progression of myopia in children with non-diseased eyes, who at the initiation of treatment are 8-12 years of age and have a refraction of -0.75 to -4.00 diopters (spherical equivalent) with ≤ 0.75 diopters of astigmatism. The lens is to be discarded after each removal.
**Based on a clinical study in which participants were between the ages of 8 and 12 at initial fit.
†Compared with a standard single vision, 1 day lens over a three-year period.
‡ MiSight® 1 day is defined as orders and includes MiSight® 1 day product(s) sold and distributed by CooperVision in the US.
$ Amount is defined as weight. Net plastic neutrality is established by purchasing credits from Plastic Bank®. A credit represents the collection and conversion of one kilogram of plastic that may reach or be destined for waterways. CooperVision purchases credits equal to the weight of plastic in MiSight® 1 day orders in a specified time period. MiSight® 1 day plastic is determined by the weight of plastic in the blister, the lens, and the secondary package, including laminates, adhesives, and auxiliary inputs (e.g. ink).
1. Chamberlain P, et al. A 3-Year Randomized Clinical Trial of MiSight® Lenses for Myopia Control. Optom Vis Sci. 2019;96(8):556-7. 2. Tideman JW, et al. Association of Axial Length With Risk of Uncorrectable Visual Impairment for Europeans With Myopia. JAMA Ophthalmol. 2016;134(12):1355-63.
2. CVI Data on file, 2022.