MiSight® Today.
Anything Tomorrow.

MiSight® Today.
Anything Tomorrow.
Correct and help protect your child's blurry vision from getting worse as they grow, with MiSight® 1 day. *†‡1,2
If your child has trouble seeing far away (nearsightedness, known as myopia), you can correct AND help protect their vision from getting worse as they grow. Meet MiSight® 1 day soft contact lenses specifically designed for myopia control and FDA approved* to slow the progression of myopia in children ages 8–12 at the initiation of treatment. §1
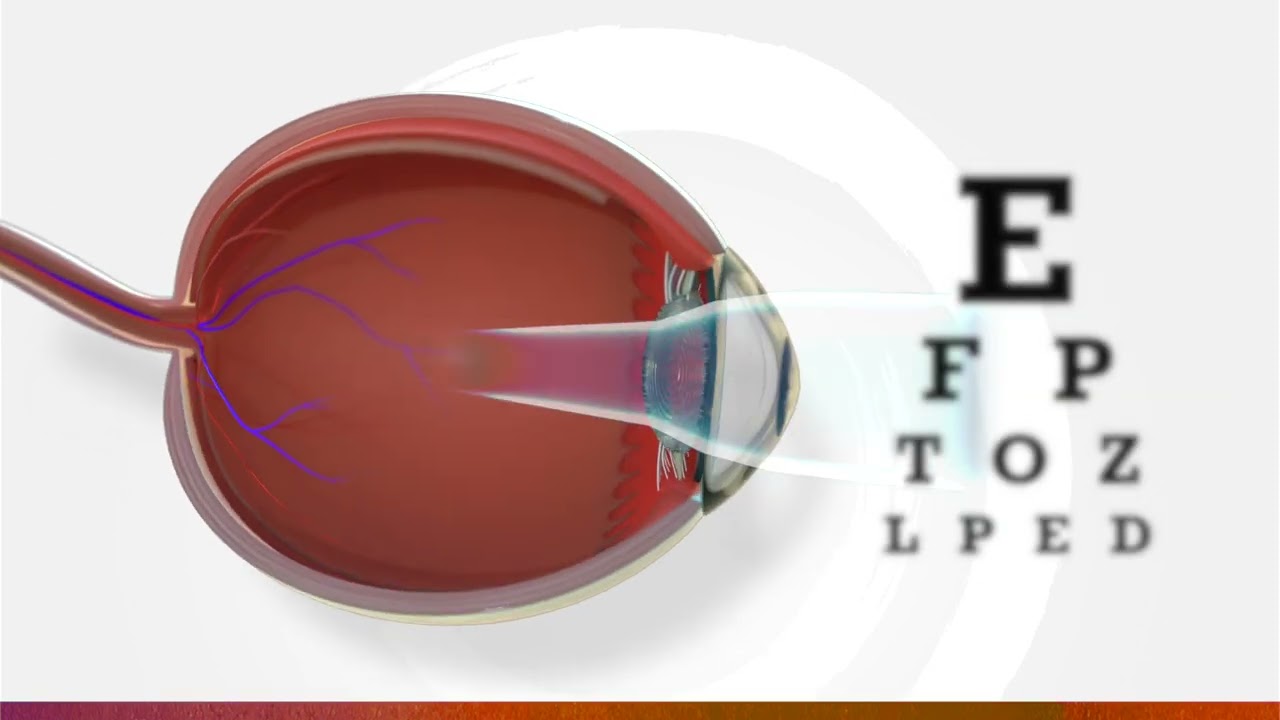
What is myopia (nearsightedness)?
The eyes of a child with nearsightedness grow faster and longer than they should, causing far away objects to appear blurry. If untreated, the eye becomes weaker and vision often gets worse as the child grows older.
See through your child’s eyes.
Our simulator shows how vision changes with different levels of nearsightedness.
How nearsighted is your child?

There’s more to MiSight® 1 day contact lenses than meets the eye.

The first and only FDA-approved* soft contact lenses proven to slow myopia progression in children, aged 8-12 at the initiation of treatment.§1

On average, age-appropriate children wearing MiSight® 1 day contact lenses progressed less than -1.00D over 6 years.|3

Children as young as 8 years old can confidently apply and remove MiSight® 1 day lenses on their own.¶1

Children wearing MiSight® 1 day achieved better than 20/20 vision across all visits over a 6-year period.**1,3
Hear what real MiSight® 1 day patients have to say.
Take the next step.
Myopia Blog
By prescription only. Results may vary. Please reference the Patient Information Booklet for a complete listing of Indications and Important Safety Information.
‡‡ Available on purchases between 1/1/26-6/30/26. Must submit rebate form within 60 days of lens purchase.
† ActivControl® technology in MiSight® 1 day contact lenses slows axial length elongation and corrects refractive error for age-appropriate children.
‡ MiSight® 1 day shows sustained slowing of eye growth over time on average. While eyes are still growing; children fit ages 8-12 and followed for 6-years. n=40.
§ Compared to a single vision 1 day lens over a 3-year period.
| Fitted at 8-12 years of age at initiation of treatment.
¶ At initial dispense 66/67 children successfully fit with MiSight® 1 day aged 8-12 were able to handle their lenses.
** VA (LogMAR) > 6/6 (20/20) at all visits from dispensing to 6-year visit.
†† By prescription only, per doctor’s discretion. Eye Exam may be required. Free trial does not include eye exam or fitting fees. Offer valid with participating eye care practitioners in the U.S. only.
References:
1 Chamberlain P, et al. A 3-year randomized clinical trial of MiSight® lenses for myopia control. Optom Vis Sci. 2019; 96(8):556-567.
2 Chamberlain P et al. Long-Term Effect of Dual-Focus Contact Lenses on Myopia Progression in Children: A 6-year Multicenter Clinical Trial. Optom Vis Sci 2022 In Press.
3 Chamberlain P, Arumugam B, Jones D et al. Myopia Progression in Children wearing Dual-Focus Contact Lenses: 6-year findings. Optom Vis Sci 2020;97(E-abstract): 200038.